Abstract
Airway inflammation and remodeling cause respiratory impairment in Cystic Fibrosis (CF). We aim to investigate the immunological changes in human end-stage CF lungs by tissue RNA transcriptional profiling to improve insight in the inflammatory component of the disease.
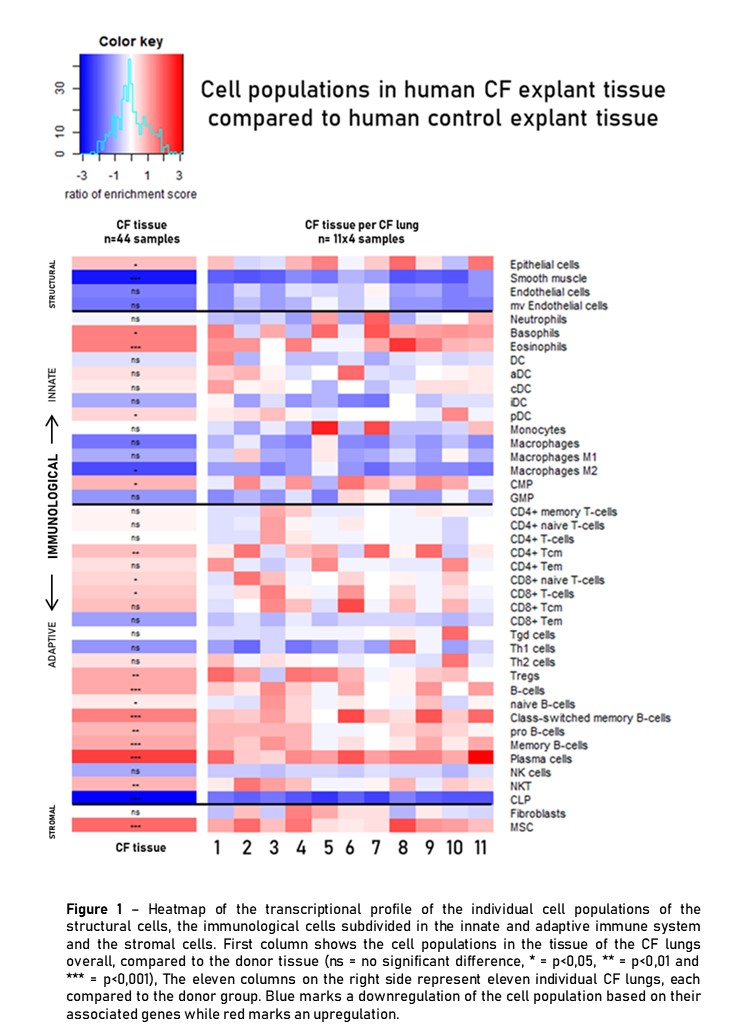
Bulk RNA profiling (Ion Ampliseq Tech) was performed on frozen explant CF (n=11) and unused donor lungs (n=11)(4 randomly selected samples/lung; n=88). Cell type enrichment analysis was performed in R with xCell (Aran D. Meth Mol Biol 2020) from gene expression data for immune/stromal cell types. Results of individual cell populations are presented as a heatmap overall and per CF lung (Fig1).
Epithelial cells (EC) were up- and smooth muscle cells (SMC) downregulated in CF vs. donor lungs. Upregulated innate immune cells included eosinophils and basophils, whereas macrophages were downregulated and neutrophils were overall similar, potentially due to a pre-apoptotic state. A pronounced upregulation of the adaptive immune cells is present including CD4+ and CD8+ T-cells, Tregs, B-cells and plasma cells, confirming the chronic inflammatory state. Stromal cells showed no changes for fibroblasts but mesenchymal stem cells (MSC) responding to chronic lung injury, were upregulated.
In end-stage CF the chronic inflammatory state of the disease is reflected by a pronounced adaptive immune response in the lung accompanied by structural alterations in both EC, SMC and an increase of MSC.