Abstract
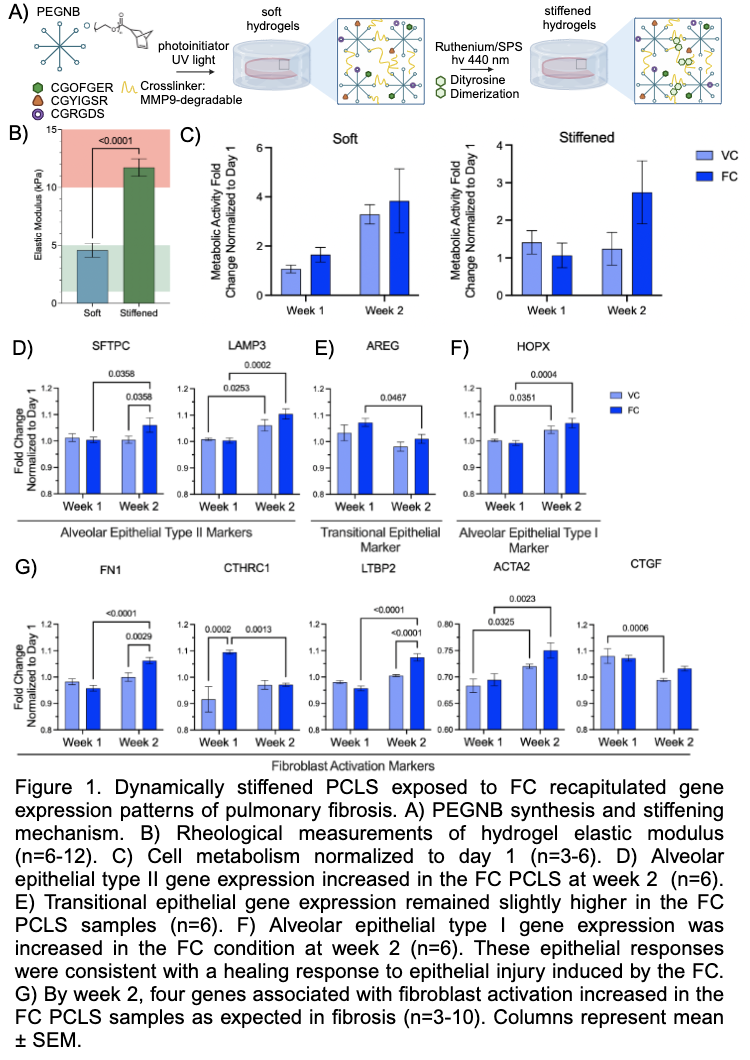
Precision cut lung slices (PCLS) represent a versatile, physiologically relevant tool that facilitates advancements in pulmonary research and drug development. Traditionally, PCLS are limited by short term viability. To extend the utility of PCLS fibrosis models, healthy murine-derived PCLS were embedded in tunable stiffness poly(ethylene glycol) norbornene (PEGNB) hydrogels. This embedding process preserved PCLS viability and enabled dynamic stiffening with pro-fibrotic biochemical signal exposure (fibrosis cocktail; FC). Soft and stiffened hydrogels had elastic modulus values of 4.58 ± 0.61 kPa and 11.72 ± 0.75 kPa, which respectively replicates the elastic modulus of healthy and fibrotic lung tissue. After two weeks in culture, FC samples demonstrated higher cell metabolism. In stiffened FC PCLS, several genes associated with epithelial differentiation (SFTPC, LAMP3, AREG, HOPX, AGER) and fibroblast activation (CTGF, COL1A1, CTHRC1, FN1, LTBP2, ACTA2) were upregulated, reflecting repair and fibrotic responses. For instance, fibroblast responses matched expected trends in FC-exposed PCLS. Subsequent experiments will use human PCLS and test different drugs to assess potential therapeutic benefits. Extending PCLS culture can more accurately model chronic disease progression, provide valuable insight into repair and fibrotic mechanisms, cell-specific responses, and may identify novel anti-fibrotic drugs with higher clinical success.