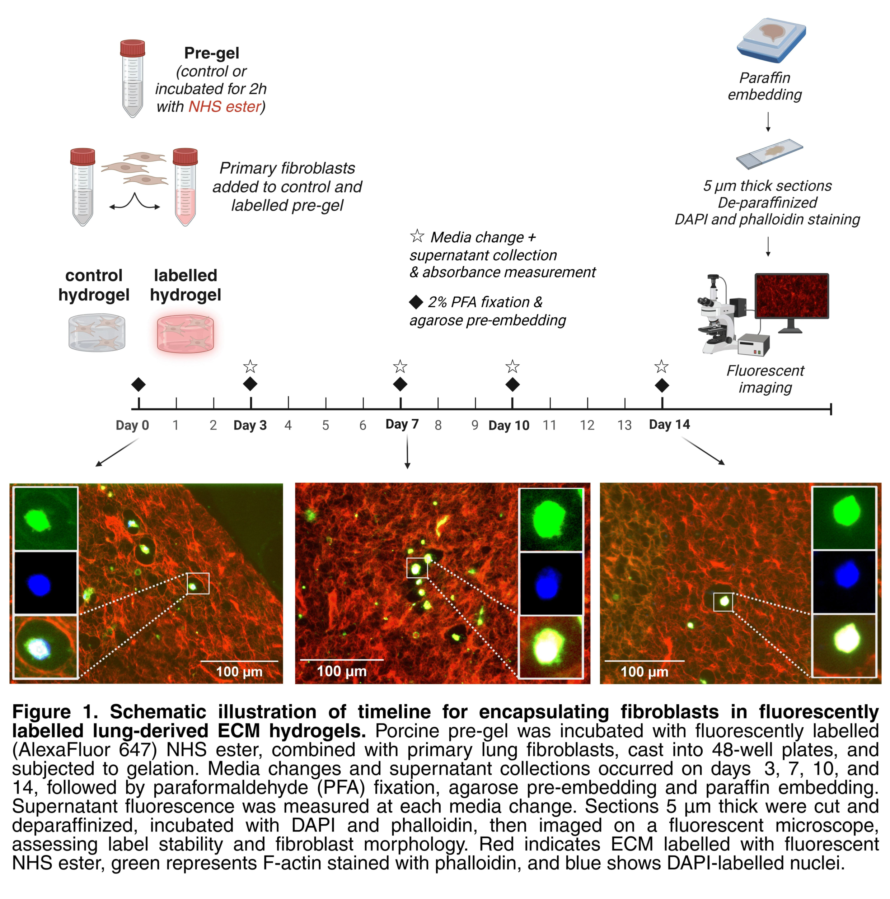
Abstract
Understanding extracellular matrix (ECM) dynamics is essential for advancing research in fibrosis and tissue repair and regeneration. The ECM provides structural and biochemical support to cells, with fibroblasts playing a key role in its production and modification. One in vitro model system that enables investigation of cell-ECM interactions and ECM remodelling is 3D native lung ECM-derived hydrogels, in which fibroblasts can be encapsulated and maintained. Current ECM visualization methods label total or specific ECM proteins at endpoints and do not distinguish between original ECM and newly deposited ECM resulting from fibroblast activity. To address this, we implemented an N-hydroxysuccinimide (NHS) ester-based fluorescent labelling as a novel method of visualizing ECM proteins in porcine lung ECM-derived hydrogels prior to gelation and fibroblast encapsulation (Figure 1). We optimized label concentration, pH, and number of washing steps needed to remove unbound label. We then confirmed label stability over 14 days using fluorescence microscopy on formalin-fixed and paraffin-embedded sections (Figure 1). Embedded primary lung fibroblasts remained viable over 14 days in labelled hydrogels. This method offers a promising approach for universal ECM protein labelling and could be extended to other model systems for studying ECM turnover in health and disease.